Cho 13g Magie tác dụng với axit clohiđric(HCL) a. Tính khối lượng muối magie clorua (MgCl2) thu được? b. Nếu dùng toàn bộ lượng chất khí vừa sinh ra ở phản ứng trên để khử đồng (ll) oxit ở nhiệt độ cao. Tính khối lượng kim loại tạo thành
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.


a) $n_{Mg} = \dfrac{4,8}{24} = 0,2(mol)$
$Mg + 2HCl \to MgCl_2 + H_2$
Theo PTHH :
$n_{HCl} = 2n_{Mg} = 0,4(mol) \Rightarrow m_{HCl} = 0,4.36,5 = 14,6(gam)$
b)
$n_{MgCl_2} = n_{Mg} = 0,2(mol) \Rightarrow m_{MgCl_2} = 0,2.95 = 19(gam)$
\(n_{Mg}=\dfrac{4,8}{24}=0,2\left(mol\right)\\ Mg+2HCl\rightarrow MgCl_2+H_2\\ 0,2.......0,4........0,2.........0,2\left(mol\right)\\ a.m_{HCl}=0,4.36,5=14,6\left(g\right)\\ b.m_{MgCl_2}=0,2.95=19\left(g\right)\)

a) \(PTHH:Mg+2HCl\rightarrow MgCl_2+H_2\)
b) \(n_{Mg}=\dfrac{4,8}{24}=0,2mol\)
\(n_{HCl}=2.n_{Mg}=0,2.2=0,4mol\)
\(\Rightarrow m_{HCl}=n.M=0,4.36,5=14,6g\)
c) \(n_{H_2}=n_{Mg}=0,2mol\)
Thể tích khí hidro sinh ra (ở đktc):
\(V_{H_2}=0,2.24,79=4,958l.\)

a.b.\(n_{Mg}=\dfrac{3,6}{24}=0,15mol\)
\(Mg+2HCl\rightarrow MgCl_2+H_2\)
0,15 0,3 0,15 ( mol )
\(V_{H_2}=0,15.22,4=3,36l\)
\(m_{HCl}=0,3.36,5=10,95g\)
c.\(n_{H_2}=0,15.60\%=0,09mol\)
\(Ag_2O+H_2\rightarrow\left(t^o\right)2Ag+H_2O\)
0,09 0,18 ( mol )
\(m_{Ag}=0,18.108=19,44g\)
\(a.n_{Mg}=\dfrac{3,6}{24}=0,15\left(mol\right)\\ Mg+2HCl\rightarrow MgCl_2+H_2\\ TheoPT:n_{H_2}=n_{Mg}=0,15\left(mol\right)\\ \Rightarrow V_{H_2}=0,15.22,4=3,36\left(l\right)\\ b.TheoPT:n_{HCl}=2n_{Mg}=0,3\left(mol\right)\\ \Rightarrow m_{Mg}=0,3.36,5=10,95\left(g\right)\\ c.n_{H_2\left(pứ\right)}=0,15.60\%=0,054\left(g\right)\\ H_2+Ag_2O-^{t^o}\rightarrow2Ag+H_2O\\ n_{Ag}=2n_{H_2}=0,108\left(mol\right)\\ \Rightarrow m_{Ag}=0,108.108=11,664\left(g\right)\)

nMg = 3,6 : 24 = 0,15 (mol)
pthh : Mg + 2HCl --> MgCl2 + H2
0,15-----------> 0,15 --->0,15 (mol)
mMgCl2 = 0,15 . 95 = 14,25 (mol)
VH2 (đkc)= 0,15. 24,79 = 3,718(l)
\(n_{Mg}=\dfrac{3,6}{24}=0,15mol\)
\(Mg+2HCl\rightarrow MgCl_2+H_2\)
0,15 0,3 0,15 0,15
\(m_{MgCl_2}=0,15\cdot95=14,25g\)
\(V_{H_2}=0,15\cdot22,4=3,36l\)

\(a.Mg+2HCl\rightarrow MgCl_2+H_2\\ n_{H_2}=\dfrac{44,8}{22,4}=2\left(mol\right)\\ n_{HCl}=2n_{H_2}=4\left(mol\right)\\ \Rightarrow m_{HCl}=4.36,5=146\left(g\right)\\ c.n_{MgCl_2}=n_{H_2}=2\left(mol\right)\\ \Rightarrow m_{MgCl_2}=2.95=190\left(g\right)\)

a.\(n_{Al}=\dfrac{5,4}{27}=0,2\left(mol\right)\)
\(2Al+6HCl\rightarrow2AlCl_3+3H_2\uparrow\)
0,2 0,3 ( mol )
\(V_{H_2}=0,3.22,4=6,72\left(l\right)\)
b.\(n_{CuO}=\dfrac{48}{80}=0,6\left(mol\right)\)
\(CuO+H_2\rightarrow\left(t^o\right)Cu+H_2O\)
0,6 > 0,3 ( mol )
0,3 0,3 ( mol )
\(m_{Cu}=0,3.64=19,2\left(g\right)\)
c.\(2H_2+O_2\rightarrow\left(t^o\right)2H_2O\)
0,3 0,15 ( mol )
\(V_{kk}=V_{O_2}.5=\left(0,15.22,4\right).5=16,8\left(l\right)\)

\(n_{Mg}=\dfrac{6}{24}=0,25\left(mol\right)\\
pthh:Mg+H_2SO_4->MgSO_4+H_2\)
0,25 0,25 0,25 0,25
\(m_{MgSO_4}=0,25.120=30\left(g\right)\)
\(n_{Fe_2O_3}=\dfrac{24}{160}=0,15\left(mol\right)\\
pthh:Fe_2O_3+3H_2\underrightarrow{t^o}2Fe+3H_2O\)
LTL : \(\dfrac{0,15}{1}>\dfrac{0,25}{3}\)
=> Fe dư , H2 hết
=> \(m_{Fe}=\dfrac{1}{6}.56=\approx9,3\left(g\right)\)

a)
\(PTHH:Mg+2HCl->MgCl_2+H_2\)
2<------4<----------2<---------2 (mol)
b)
\(n_{H_2\left(dktc\right)}=\dfrac{V}{22,4}=\dfrac{44,8}{22,4}=2\left(mol\right)\)
\(m_{HCl}=n\cdot M=4\cdot\left(1+35,5\right)=146\left(g\right)\)
c)
\(m_{MgCl_2}=n\cdot M=2\cdot\left(24+71\right)=190\left(g\right)\)
\(n_{H_2}=\dfrac{V}{22,4}=\dfrac{44,8}{22,4}=2\left(mol\right)\)
\(\text{a)}Mg+2HCl\rightarrow MgCl_2+H_2\)
\(2mol\) \(1mol\) \(1mol\)
\(4mol\) \(2mol\) \(2mol\)
\(b)m_{HCl}=n.M=4.36,5=146\left(g\right)\)
\(c)m_{MgCl_2}=n.M=2.95=190\left(g\right)\)

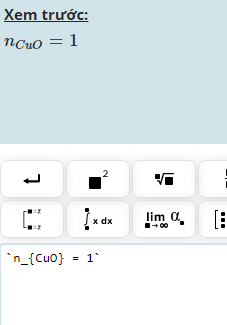

\(n_{Mg}=\dfrac{13}{24}=0,54mol\)
\(Mg+2HCl\rightarrow MgCl_2+H_2\)
0,54 0,54 ( mol )
\(m_{MgCl_2}=0,54.95=51,3g\)
\(CuO+H_2\rightarrow\left(t^o\right)Cu+H_2O\)
0,54 0,54 ( mol )
\(m_{Cu}=0,54.64=34,56g\)